- Home
- Applications
- Measure Cell Migration and Chemotaxis in Real Time
- Incucyte® Chemotaxis Assay

Incucyte® Applications
Chemotactic Migration Assays for Live-Cell Analysis
What is chemotaxis?
What is chemotaxis?
Chemotaxis is the directional movement of cells in response to a chemical stimulus and is an essential component of immune responses, tumor metastasis, wound healing and blood vessel formation. Cell-based assays for chemotaxis are critical to further understanding these biological processes.

4th Edition! Live-Cell Analysis Handbook
A guide to real-time live-cell imaging & analysis

Application Note:
Gain additional insights into the mechanism of immune cell killing by combining live cell analysis and flow cytometry into a single workflow
Download Your CopyIntroducing the Incucyte® Chemotaxis Assay
A novel, integrated solution for real-time visualization and automated analysis of chemotactic cell migration in a 96-well format – all inside your tissue culture incubator.
Chemotaxis Cell Migration Quick Guide

Key Advantages
Key advantages of the Incucyte® Chemotaxis Assay
- Visualize cell migration in real time with fully automated analysis
- Highly reproducible 96-well kinetic assays suitable for screening
- Low cell usage ideal for rare, expensive and primary cell populations
- Relevant surface-mediated migration and detailed phenotypic insight
Download the Incucyte® Chemotaxis Assay Application Note
Common limitations of existing cell-based assays for chemotaxis
- Inability to visualize cell migration in real time (e.g., Boyden chamber transwells)
- Low throughput, technically challenging protocols (e.g., microfluidic devices)
- Lack of integrated quantitation
Visualize cell migration in real time with fully automated analysis
- Monitor every cell in your experiment, assess morphology and gain phenotypic insight from images and movies
- Validated for use with adherent and non-adherent cell types including immune, cancer and vascular cells
- Measure label-free or labeled cell migration without fixing, staining or cell scraping steps
Visualize and quantify chemotactic migration in your choice of cells. The Incucyte® Chemotaxis Assay has been validated with many adherent and non-adherent cell types including immune, cancer and vascular cells, and a wide range of chemoattractants, growth factors and biomatrix coatings. Watch the demonstration videos below.
Measuring Chemotaxis of HT-1080 cells
Measuring Chemotaxis of T cells
Watch more videos
Measuring Chemotaxis of HUVEC cells
Measuring Chemotaxis of Neutrophils
Highly reproducible 96-well kinetic assays suitable for screening
- Measure chemotactic cell migration in up to six 96-well plates at once (576 wells of data at every time point)
- Setup and walk away – fully automated image based quantification
- Compatible with standard automation and liquid dispensing devices
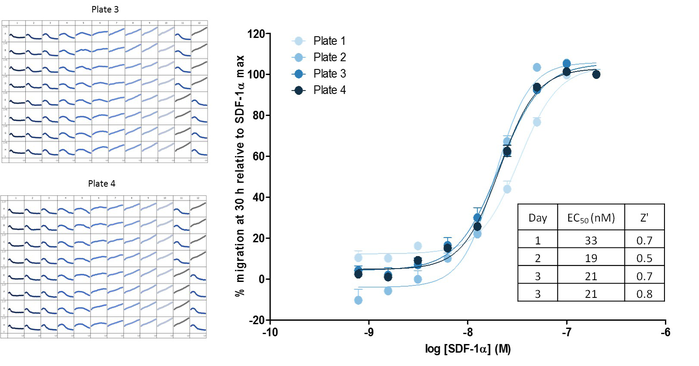
Measuring chemotaxis using robust 96-well assays. Representative 96-well microplate graphs showing Jurkat migration towards the chemoattractant SDF-1α to illustrate inter-plate reproducibility (serial dilutions of chemoattractant across the plate). Z’ values ranged from 0.5 to 0.7 for four replicate plates over three days. Corresponding concentration-dependent response curves to SDF-1α provided reproducible measurements of SDF-1α potency (EC50 value range 19 to 33 nM) within and between days.
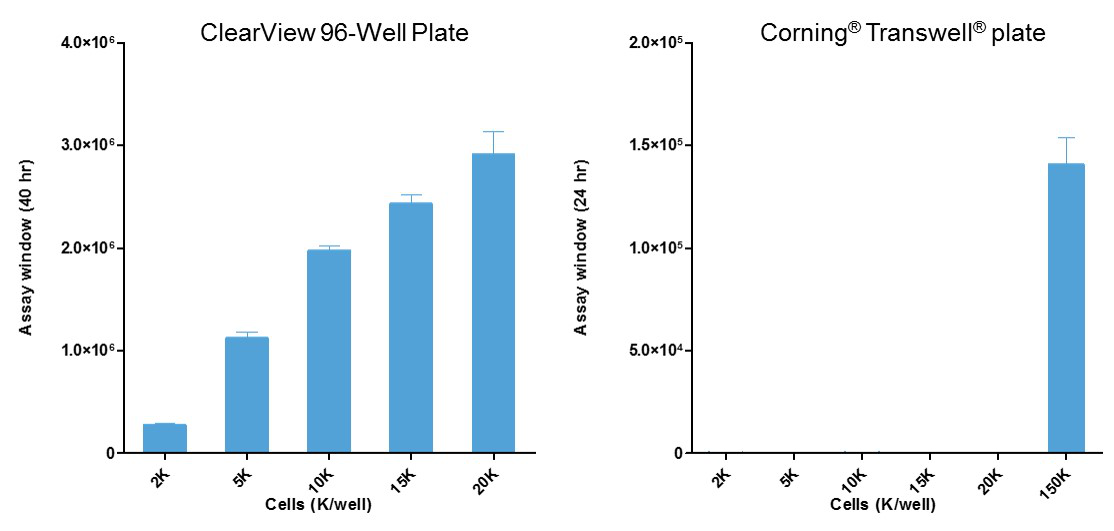
Low cell usage ideal for rare, expensive and primary cell populations
- Use 10- to 100-fold fewer cells than standard transmembrane (e.g., Boyden chamber) approaches
Relevant surface-mediated migration and detailed phenotypic insight
- Investigate cell migration on biologically relevant surfaces
- Monitor morphological changes, cell-cell interactions and collective cell migration
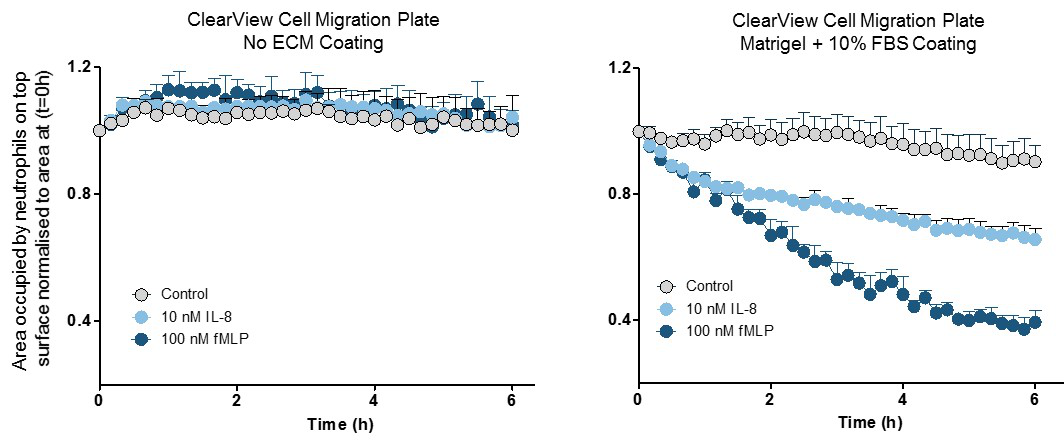
Measure relevant surface contact-mediated cell migration. The low pore density of the Clearview membrane ensures that cells must migrate across the biologically relevant surface towards the chemoattractant. Neutrophils seeded on an uncoated Clearview membrane were unable to migrate towards the chemoattractants IL-8 and fMLP (left); however, those on Matrigel-coated membranes showed clear chemotactic profiles (right). These data suggest that integrin and/or cell surface receptor interactions with the substrate play a key role in neutrophil chemotaxis in this model. In contrast, no coatings were required for neutrophil migration studies using the Corning® Transwell® consumable (data not shown), suggesting that active migration of neutrophils across the Transwell® filter is absent in the Corning® Transwell® system.
FAQs
Chemotaxis FAQs
Membrane-based transwell systems for chemotaxis assays are capable of maintaining chemoattractant gradients for a few hours, but beyond that they collapse, leading to equimolar amounts of chemoattractant on both sides of the membrane. ClearView 96-Well Chemotaxis Plates rely on 96 tiny pores (8 µm in diameter) to set up and maintain the chemoattractant gradient for 72 hours. Since chemotaxis migration assays rely on chemical gradients to differentially induce migratory behavior in cells, maintaining the chemoattractant gradient is essential.
Yes, rather than just citing the reduction of cells on the surface of the membrane, the IncuCyte™ ZOOM can provide real-time analysis of the movement of cells through the pores to the underside of the well. It simultaneously provides live-cell analysis of both surfaces of the insert, enabling morphological analysis of the cells before, during, and after migrating through the pores.
Chemotaxis assays analyze cell migration in response to a chemoattractant, a process that guides cell trafficking in vivo. Examples of chemotaxis assay applications include the analysis of chemotactic migration across substrate surfaces, chemotactic invasion through 3-D biomatrix gels, and chemotactic transendothelial migration of leukocytes (extravasation or diapedesis). Studying chemotaxis requires the generation of chemotactic gradients, as cells travel from areas with one concentration to areas of another (high concentration to low concentration, or low concentration to high concentration). Some chemotaxis assays rely on membranes to maintain these chemoattractant gradients, but those gradients are unstable and they rapidly collapse. ClearView 96-Well Chemotaxis Plates enable gradient maintenance for at least 72 hours -- Something other chemotaxis assay systems can’t claim.
Learn more about the ClearView method for chemotaxis assays
Leukocyte extravasation is the process by which leukocytes in the circulation stop rolling along vascular endothelium and migrate across the endothelium in response to inflammation. In order for any system to accurately recapitulate transendothelial migration, it must first involve the generation of a monolayer of endothelial cells over top of the pore-based insert. Once confluence of the endothelial cells is established, the leukocytes can be carefully added to the culture, where they will extravasate between the endothelial cells in response to the chemoattractant in the reservoir.
View the IncuCyte™ transendothelial migration assay protocols
The most common effector cells used in the study of trans endothelial migration of leukocytes in vitro include primary T cells, neutrophils, and Jurkat cells. The most commonly used cells to recapitulation the endothelial cell layer are HUVECs. Other cells not listed here that exhibit the ability to extravasate through the endothelium may work with this system, but the protocol – including the choice of endothelial monolayer – will need to be experimentally determined.
While immune cells are non-adherent, their interaction with ECM on the substrate is essential for their migration. Immune cells do not migrate on uncoated surfaces, but can readily migrate in the presence of appropriate ECM proteins. Examples of substrate coatings that are compatible with immune cell chemotaxis include ICAM-1, fibronectin, and Matrigel®/FBS.
Ordering Info
Incucyte® Chemotaxis Analysis Software Module
An add-on software module for the Incucyte® Live-Cell Analysis System. Analyze label-free and fluorescently-labeled chemotactic cell migration images acquired using the Incucyte® Clearview Plate. This software is required for analyzing chemotaxis using the Incucyte® Live-Cell Analysis System.
Incucyte® Clearview 96-Well Plate for Chemotaxis
The Incucyte® Clearview 96-Well Plate provides an optically clear surface for label-free imaging and analysis of chemotactic cell migration within the Incucyte® Live-Cell Analysis System. The plates are required for analyzing chemotaxis using the Incucyte® Live-Cell Analysis System.
Tech Resources
Technical Resources
Documentation
- AACR 2016 Poster: Chemotactic Cell Migration
- Incucyte® Chemotaxis Application Note
- Incucyte® Chemotaxis Quick Start Guide
Posters
- ELRIG 2015: Incucyte® Chemotaxis
- AAI 2015: A New Technology for In Vitro Chemotaxis Assays
- AAI 2016: Kinetic Measures of Macrophage Chemotaxis
- CIMT 2016: Quantitative live-cell imaging assays for immunotherapy: chemotaxis, immune cell killing & phagocytosis