In our fight against viral pneumonia, the “Mighty Macrophage” packs a punch
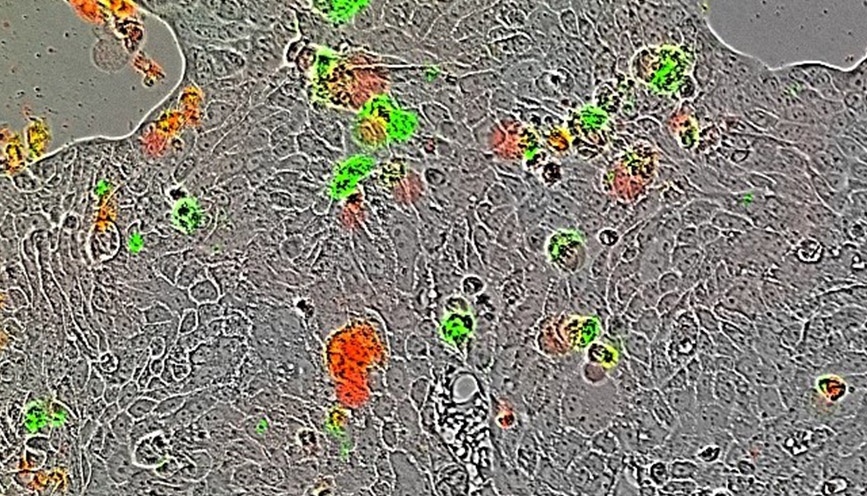
Calu-3-lun carcinoma cells infected with Influenza A (PR8), treated with human recombinant Macrophage Migration Inhibitory Factor (MIF). IncuCyte Annexin V used for viability assessment. – Image courtesy of Dr. Candice Smith
By Jill Granger, M.S., Technical Writer, Product Management, Sartorius
It’s hard not to tap into the anxiety as the COVID-19 pandemic travels the globe and impacts our daily lives. But now’s the time for some hopeful perspective, remembering that immune cells have our back. Among our arsenal is the “Mighty Macrophage”, which can be either friend or foe–depending on how well it plays in the sandbox.
Macrophages have always been a favorite of immunologists. With their bigger-than-life presence under the microscope, who can resist their charm as they gobble up pathogens, and clear erythrocytes and cellular debris like giant cellular garbage trucks? When they behave well, they are mighty defenders, working as part of our innate immune system to sound the alarm and clear infections. But like anyone’s unruly relatives at a family wedding, they can sometimes consume too much and cause an uproar, leaving the wedding hall in shambles. At other times, they may be content to “just hang-out in the corner” and put a damper on the party. Indeed, controlling this duality of “macrophage behavior issues”, or plasticity, may be the key to reducing the mortality from infectious disease.
Our innate immune system provides the first line of defense against a foreign invaders. As viruses or bacteria enter the lung, the innate immune response is initiated. Alveolar macrophages signal the alarm as one of the first responders, phagocytosing intruders and presenting their antigens, secreting cytokines and chemokines to initiate a pro-inflammatory responses and recruitment of leukocytes such as neutrophils. Antiviral proteins, such as interferons, are produced to stop viral replication. In the best case scenario, the infection is overcome by an appropriately-gauged immune response that destroys and eliminates the pathogen. This response is normally quelled by an anti-inflammatory response from another subset of macrophages, which turn off the body’s response as the infection subsides. However, the ability to keep things in check can sometimes go awry. In some people, severe pneumonia may arise following bacterial and viral infections. Known as Acute Respiratory Distress Syndrome (ARDS), the proinflammatory immune response goes out of control like a wildfire. Inflammatory cytokines and chemokines are overproduced, resulting in lung tissue damage and fluid accumulation in the lungs, impeding life-sustaining oxygenation of the organs. This can give rise to complications such as organ failure and even death.
Controlling this collateral damage when cells and inflammatory mediators “behave badly” has been subject of investigation for reducing ARDS mortality and organ failure from influenza. In a recent JCI Insight publication, Dr. Candice Smith and colleagues from Dr. Daniel Goldstein’s lab at the University of Michigan attempted to better understand macrophage regulation in a mouse model of influenza (IAV, strain PR8). They examined the role of Macrophage Migration Inhibitory Factor (MIF), a cytokine protein that can regulate the innate immune response through a variety of means, including inhibition of apoptosis, activation of macrophages, and stimulation of NLRP3 inflammasome. Up to this point, the role of MIF in influenza infection had not been characterized. Their studies revealed that MIF impairs the anti-viral immunity of the host, giving rise to increased inflammation during influenza infection in their mouse model of influenza.
I touched base with Dr. Goldstein to learn more their findings on the role of MIF in the immune response to viral infection, a discovery which has important implications in search for new anti-viral therapeutics:
What key findings lead you to this important discovery about MIF?
“My collaborator Dr. Richard Bucala (Rick) who was a colleague of mine during my 16 years at Yale School of Medicine, is the leading authority on MIF. His laboratory has found that MIF plays roles in many diseases including rheumatological, vascular and host defense. Yet the role of MIF was strangely undefined. This seem like a gap in knowledge that needed to be filled. Rick inspired me to examine this.”
How did the use of live-cell analysis aid in your determination that MIF may be enhancing inflammation during infection by necrosis during IAV infection?
“We use fluorescently tagged influenza virus to investigate how MIF controlled viral infection. We found that MIF promoted viral spread.”
Could individual differences in an individual’s basal gene expression of MIF play a role in the severity of illness that we see with viral infections, such as COVID-19? How might MIF levels be altered as in response to age and pre-existing conditions where inflammation may already be present, prior to infection with a virus?
“Yes, this is a great point. Rick has found that the 5′ promoter of the MIF gene contains a microsatellite repeat (CATT)5–8 that regulates transcription, that shows that high MIF levels are detrimental for some viral infections, like West Nile Infection. MIF could be regulated by aging and MIF could potentially act as a marker of risk for viral infection. How MIF is involved in COVID-19 is unknown, but potentially very relevant.”
How might targeting MIF aid in reducing the mortality of particularly susceptible individuals?
“It is possible that MIF acts as a “bad player” during respiratory viral infection. Blocking it or the pathway it acts upon could increase viral control, reduce inflammation and increase survival in susceptible populations.“
This study shows the value of using a variety of techniques to closely examining the delicate balance between pro- and anti-inflammatory responses to infection that, when disrupted, can lead to disastrous consequences. Understanding the regulation of macrophage function can provide additional, invaluable details and insights for the identification of possible new therapeutic targets to combat viral infection. In our current battle against a global pandemic, we need to make sure our immune cell friends are in our corner and behaving well, because right now, we could use all the friends we can get.
This blog post is dedicated to the heroic efforts of all researchers and medical staff around the world who are tirelessly combatting infectious disease, especially those working on COVID-19 pandemic. I applaud your efforts and thank you all for your dedication.
For further reading on this topic:
Learn more about our advanced tools for evaluating viral infections and host immune responses
Smith CA et al. Macrophage migration inhibitory factor enhances influenza-associated mortality in mice. JCI Insight. 2019 Jul 11;4(13).
Visit the website for the Goldstein Lab at the University of Michigan Medical Schools
Related Publications:
Mosser DM and Edwards JP. Exploring the full spectrum of macrophage activation. Nat Rev Immunol. 2008 Dec;8(12):958-69. Review. Erratum in: Nat Rev Immunol.2010 Jun;10(6):460 Read More
Huang X, Xiu H, Zhang S, Zhang G. The Role of Macrophages in the Pathogenesis of ALI/ARDS. Mediators Inflamm. 2018;2018:1264913. Published 2018 May 13 Read More
Cheung CY, et al. Cytokine responses in severe acute respiratory syndrome coronavirusinfected macrophages in vitro: possible relevance to pathogenesis. J Virol. 2005 Jun;79(12):7819-26 Read More
Discover more about IncuCyte® applications for analysis of macrophages
Discover more about IncuCyte® applications for immunology
Read more about how scientists are using IncuCyte® for Viral Research
Credits:
Dr. Daniel R Goldstein
Eliza Maria Mosher Collegiate Professor in Internal Medicine
Research Professor, Institute of Gerontology
Professor Microbiology and Immunology
Director, Michigan Biology of Cardiovascular Aging
University of Michigan
Ann Arbor, MI USA
https://mboca.medicine.umich.edu
Twitter: @LabGoldstein
Dr. Candice Smith
Environmental Chemical Laboratory Scientist
Oklahoma Department of Environmental Quality
Oklahoma City, OK USA
candicas@umich.edu



